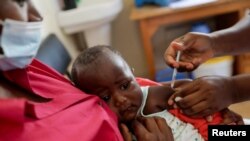
WHO endorsed the Malaria vaccine last fall as a “ historic ” breakthrough in the fight against malaria, but the Gates Foundation told AP this week it will no longer financially support the shot.
The malaria vaccine has “a much lower efficacy than we would like,” said Philip Welkhoff, the Gates Foundation's director of malaria programs.
Explaining its decision to end support after spending more than $200 million and several decades getting the vaccine to market, Welkhoff said the shot is relatively expensive and logistically challenging to deliver.
“If we’re trying to save as many lives with our existing funding, that cost-effectiveness matters,” Welkhoff added.
Some scientists say they’re mystified by that decision, warning it could leave millions of African children at risk of dying from malaria as well as undermine future efforts to solve intractable problems in public health.
“It’s not the greatest vaccine in the world, but there are ways of using it that could have a big impact,” said Alister Craig, dean of biological sciences at Liverpool School of Tropical Medicine.
The vaccine, sold by GlaxoSmithKline (GSK) as Mosquirix, is about 30% effective and requires four doses.
GSK says it can only produce about 15 million doses per year until 2028. But WHO estimates that to protect the 25 million children born in Africa every year, at least 100 million doses might be needed annually. Although there are plans to transfer the technology to an Indian drugmaker, it will be years before any doses are produced.
“We're supporting the roll-out via the Gavi funding, but we decided we would not dedicate additional direct funding to extend the supply of the vaccine,” Welkhoff said.
On Thursday, WHO and Gavi invited developing countries to apply for funding to pay for the malaria vaccine in their countries.
"If delivered to scale, the vaccine will help to prevent millions of cases of malaria, save tens of thousands of lives and ensure a brighter future for the continent,” said Dr. Matshidiso Moeti, WHO's Africa director.
The Gates Foundation's withdrawal of financial support for the malaria vaccine might unnerve others, Dr. David Schellenberg of the London School of Hygiene and Tropical Medicine, said.
“There is a risk that this could discourage others who are considering financing the malaria vaccine or even be a disincentive for people working on other vaccines,” he said.
An imperfect roll-out of the vaccine would still save lives, according to Dr. Dyann Wirth, an infectious diseases expert at Harvard University.
“We would love to have 100 million doses, but that kind of money doesn’t exist for malaria,” she said.
The Gates Foundation had done its part in bringing the vaccine to market and it is now up to countries, donors and other health organizations to ensure it is used, Wirth added.